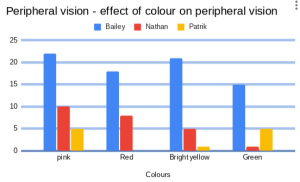
Yesterday we have been experimenting with different solutions. We dropped water droplets on a coin using pipettes. There were 4 solutions; A, B, C, D and normal water. We had to find out which one of the solutions was just normal water by putting them on coins and seeing which solution was closest to the water.
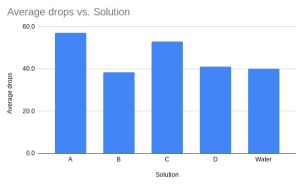
From this, the conclusion is D being water, but we didn’t have enough time to have 3 tries on every one of them, so it may be incorrect. Each solution is different because of surface tension. Water has normal surface tension, but when you add soap to the mix, it lowers. This is because the molecules have bonds to each other and soap goes in between them. When you add salt, the opposite happens, because salt increases the bonds and strengthens them.